 |
|
|
|
|
|||||||||
|
Introduction Nickel-Cadmium batteries can save much time and money when they are used to replace other non-chargeable dry batteries. They must however, be treated in a suitable manner or they could become useless within just a few weeks. I have had many people come to me with their Camcorder and handheld radio batteries totally ruined because they do not know how to take care of them. In this article I will show you how to treat NiCad batteries so that you can use them for many hundreds of charge cycles. I will also show you some techniques to revive dead NiCad batteries. I will first present a little information about dry-batteries. I will only discuss the type AA battery size when giving figures so that you can make a meaningful comparison between the different types |
||||||
|
Safety All normal batteries have an electrolyte between plates and this stuff is quite dangerous. A battery cell with a broken case must be thrown away for the sake of safety. On Deptember the 20th 1990, the US Environment Protection Agency introduced a regulation that classified spent batteries as "Hazardous Waste" under the "Resource Conservation and Recovery Act". This means that you can get into serious shit if you just dump them. Here in Sweden most battery retailers and supermarkets have containers for disposing of spent batteries. Many other counteries have similar systems, please use them! Having got that over with, let us proceed with something we can do |
||||||
|
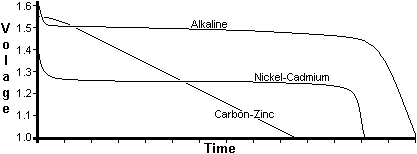
Comparisons Different batteries have different discharge properties and this can an impact upon the type you choose for your equipment. In general, it is the terminal voltage, capacity and operating temperature range that will affect your choice |
||||||
 |
||||||
|
The old Zinc-Carbon batteries have a nominal 1.5 volt from each cell but this dwindles rapidly as the cell is used. Many devices may suffer a loss of performance due to this effect. Should your equipment be designed for 12-volt (13.8-volt) operation then only nine cells are needed. Many commercial pieces of equipment (eg. Trio 2200 transciever) have battery receptacles for ten cells. One of the cell positions must be fitted with a dummy cell to allow operation from nine cells. Typical capacity = 500mA/Hour. Alkaline cells also deliver a nominal 1.5-volt per cell but the output is more constant during the cells life. There will be still be a gradual reduction in performance but two or three times the capacity of the Zinc-Carbon batteries. Again, should your equipment be designed for 12-volt (13.8-volt) operation then only nine cells are needed and one of the cell positions must again be fitted with a dummy cell to allow only nine cells to be fitted. Typical capacity = 1200mA/Hour. Nickel-Cadmium batteries only have a 1.25-volt output per cell but this is somewhat more constant than Zinc-Carbon and Alkaline cells. Ten cells would be needed to make up a 12-volt (13.8-volt) battery pack. Typical capacity = 500mA/Hour. |
|
Zinc-Carbon Batteries They are cheap. The electrolyte used to leak but today they are usually much better protected. If they should leak then they will corrode all the copper in your equipment. the corrosion will travel down wires and eat its way through Printed Circuit Boards (PCBs). At high temperatures (25 degrees or more) Zinc-Carbon batteries will give up to 25% more capacity but the shelf-life will deteriorate very rapidly. Around freezing point their shelf-life can be extended by as much as 300% so one tip is to store them in the refrigerator. Unfortunately they must be thrown away when they are exhausted. You can extend their life by up to 60% by using "Dirty-DC" to recharge them but this will also reduce the shelf-life. |
|
| ||||||||||||||||||||||||||
|
You can also cook exhausted battery cells in the oven. About 80 degrees centigrade for five to ten minutes, no more or they may explode. This technique was demonstrated on UK TV in the series "Steptoe & Son" (här i Sverige i "Albert och Herbert"). I do not reccomend that you should try to sell the cells again as new batteries! |
|
Alkaline Cells I have been told that the Dirty-DC method also works for them but I have never tried it. Alkaline cells will more readily explode than Zinc-Carbon cells so it is both pointless and risky. Cooking them in the oven does work though! Alkaline batteries will deliver more current which makes them a potential hazzard if shorted. When you next travel by air, notice how they make you semove batteries from the safety of your shaver and put them in your pocket with your metallic keys and money. Unfortunately Alkaline cells are rather expensive so it is much more cost effective to consider NiCad cells. |
|
NiCad Construction |
|
| |||||||||||||||||||||||||||||
|
Charging NiCads NiCad batteries should be charged at the 0.1C rate where C is the total capacity of the battery. For example, a 500mA/Hour battery will deliver 500mA current for one hour at 20 degrees centigrade. It should therefore be charged at 0.1 x 500 = 50mA. It should also be charged for 10 hours plus 40% = 14 hours. THIS ONLY APPLIES to a cell that has been fully dis-charged. |
|
| ||||||||||||
|
NiCads should be charged at 20 degrees Centigrade room temperature. If they are too cold or warm then they will only hold a smaller percentage of the charge given. If you have just dragged them in from the cold then allow them to warm up naturally for a couple of hours before charging. |
|
Overcharging NiCads This is what most people do. In the 60's an electric shaver manufacturer introduced a brand new shaver that was stored in a charger when not in use. The batteries were constantly charged twenty three hours and fifty minutes a day (assuming one needed the other ten minutes to shave). So what really happened to the poor old batteries? They got hot and they failed quickly. This company did for cordless shavers what king Herod did for child welfare! |
|
| ||||||||||||
|
Some batteries are advertised and sold as "quick-charge" or "fast-charge" batteries. The same rules apply, a long gentle charge is far better than a short stressful (and hot) fast charge. Quick-charge batteries may last a little longer when charged fast, but be nice to them whenever possible and they will last longer. |
|
Discharging NiCads Before you charge a cell it should ALWAYS be discharged. Each cell should be charged to about 1.00-volt to 1.05-volts. Since the terminal voltage of the cell is reasonably constant then it could be discharged with a simple resistor and then you sit and watch the terminal voltage until it falls to one volt per cell. If you have not got so much time to spare then you can make a simple discharger for battery packs. Here is an example of such a unit. |
|
| |||||||||||||
|
When the battery voltage falls to 1-volt per cell (10 cells = 10-volts) then the zener diode will not allow sufficient voltage to turn ON the darlington pair. In practice, the LED will slowly begin to dim a little before it goes out completely. Again, a slow discharge is far better than a fast discharge. Measuring this discharge time is also an indication of the true capacity of the battery NiCads will also discharge themselves, with time. In general, a normal NiCad battery will discharge itself at about 2% per day. So don't expect to charge a battery after use and expect it to be ready for your field-day six weeks later. Always discharge then recharge again before use. A self discharged battery may be allowed to drop to zero volts per cell without damage. |
|
NiCad Capacity Today there are AA sized cells that are so-called "high capacity". They function in exactly the same way but perhaps have a larger surface area with thinner materials inside the can. The same rules apply as for the ordinary NiCad batteries. You could charge them with a higher current or charge them at the same current for a proportionately longer time. NiCad batteries also change their capacity to deliver power with temperature. They also change their charge capacity with temperature as shown below. |
|
| ||||||||||||
|
A battery with a rated 500mA/Hour capacity may not in reality store the 500mA/Hour, it all depends upon how you use it. If you discharge the battery at 50mA then you may realise 580mA/Hour capacity or more. If however you discharge it at 500mA then you may only see only 450mA/Hour. |
|
Memory Effect Most people have heard of the "Memory Effect". This is where a NiCad battery has been repeatedly "topped-Up" after only a partial discharge, usually by those who have never heard of "the memory effect"!. What happens is that the battery can only be used for the part of the capacity that has been exercised. In many cases the memory effect is blamed for failure when the real cause is gross overcharging. Fully discharging to 1.0-volt per cell before charging again will prevent this from happening. If you should have a battery that genuinely suffers from the memory effect then a few FULL charge/discharge cycles should remedy this. |
|
Reviving Dead NiCad cells In some cases a NiCad cell can go down completely, but the chances of a full revival are quite good. I use two techniques together to bring dead batteries (short-circuit) back to life. Give the cell ONE good "thwack" with a hammer, square on the bottom. Be very carefull not to hit it so hard that it causes a dent. Make sure that you hit it square on the bottom with the flat part of the hammer. Charge the cell for 1/4 second at 20 times the rated capacity (10 amperes for a 500mA/Hour AA type cell). Wait a minute for it to cool down then repeat until the battery voltage comes up to 1.25-volts. Give up after about 10 - 15 attempts have failed. Conductive crystals build up between the battery plates and these make the cell a total short-circuit. These two techniques can shatter these crystals so the rest of the cell will function again. A cell may develop these metalic crystals on overcharging when the plates are stripped of coating and fine conductive hairs or crystals grow between the plates. If a cell is found to be open-circuit then the chances of revival are much lower. The application of a small current (10mA limit) at 30-volts or more may often cause the cell to begin to draw current again. Allow this to continue until the battery voltage has fallen to under 2-volts, then charge in the normal way. Cycle charge/discharge two or three times, checking the capacity at each discharge. It should be seen that the capacity increases with each charge/discharge cycle. |
||||
|
Battery Packs Using NiCads This can be a real minefield. It seems that battery packs for many household items, such as cordless drills, handheld radios and Camcorders are nicely sealed so that you cannot get into them without breaking them open. Not only that but they are so damn expensive to replace. But I suppose it is good business for companies such as Alinco, Icom, Sony and Philips. The basic problem with battery packs is that all the cells are not 100% identical. If one cell then becomes discharged before the others and you continue the discharge, then the cell that has given up first will be reverse charged by the good cells. This reverse current charging can cause weaker battery cells to become totally dead. With a little patience it is possible to neatly smash apart most battery pack cases to gain access to the individual cells. Replacement cell must have the same capacity (or more) than the others remaining cells in the pack. The memory effect has the positive effect of equalising cell capacities during normal use. Do not forget that a new replacement cell should also have the same state of charge as those remaining in the battery pack. |
||||
|
The Future ?? The future or rechargeable batteries could prove to be rather exciting. The general population want pocket telephones and other equipment that will operate for days with only one charge. The general public also try to "top-up" batteries or overcharge "... till thy kingdom come ...". Nickel Metal Hydride (NiMH) batteries could be a solution. When I have learned more about them I will update this article. |
||||